Geopolymer leaching in water and acetic acid
DOI:
https://doi.org/10.21809/rilemtechlett.2020.124Keywords:
Geopolymers; Leaching; Acid attack; Degradation; SEM-EDS.Abstract
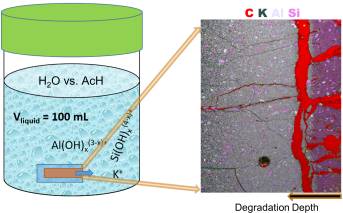
Elemental leaching of metakaolin based geopolymers was investigated by immersing hardened paste specimens in a solution. For this, pure water and 0.1 molar acetic acid solutions were replenished ten times distributed over 56 days in total. Dissolution and diffusion of the elements through and from the geopolymer paste into the surrounding solutions was investigated on cross-sections of specimens by SEM-EDS microscopy, indentation, X-ray powder diffraction analysis and measuring the eluted elements by ICP-MS when replenishing the solution over time. The presented new methodological approach thus combines the dissolution rate kinetics obtained via wet chemistry (ICP) with the complementary solid state characterisation methods to gain new insights into the complex geopolymer dissolution mechanisms.
Results indicated a relatively small leachability of geopolymers, limited only to the surface layer which is directly exposed to the aggressive solution, while the more inner parts of the geopolymer framework remain intact. Elemental maps revealed dissolution of aluminates that occurred across the outermost surface parts of the sections, while potassium leached out gradually but reached deeper inner parts. However, there was still a high portion of potassium being left bonded inside the geopolymer, even for the harsh acidic conditions, limited by the diffusion-reaction mechanism which took place within the geopolymer. The obtained experimental results represent a first approach towards feeding reactive transport numerical modeling approaches still to be developed for simulating leaching and degradation of geopolymer materials when exposed to water or acidic solutions.
Downloads
Published
How to Cite
Issue
Section
License
Authors retain copyright of the articles published in RILEM Technical Letters and grant the journal the right of first publication with open access. The work is simultaneously licensed under Creative Commons Attribution 4.0 International License (CC BY 4.0) that allows others to share and adapt the work under the following terms: 1) a proper attribution is given in a form of a reference to the original work's authorship and initial publication in RILEM Technical Letters (bibliographic record with the DOI link); 2) a link to the license is provided; 3) the changes (if any) are indicated.









