Instantaneous activation energy of alkali activated materials
DOI:
https://doi.org/10.21809/rilemtechlett.2018.78Keywords:
Activation energy, alkali activated materials, calorimetry, blast furnace slag, fly ashAbstract
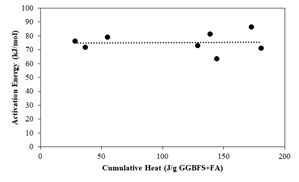
Alkali activated materials (AAM) are generally cured at high temperatures to compensate for the low reaction rate. Higher temperature accelerates the reaction of AAM as in cement-based materials and this effect is generally predicted using Arrhenius equation based on the activation energy. While apparent activation energy is calculated from parallel isothermal calorimetry measurements at different temperatures, instantaneous activation energy is typically measured using a differential scanning calorimeter. Compared to the apparent activation energy, instantaneous activation energy has minimal effects on the microstructural changes due to the variation in temperature. In this work, the evolution of activation energy was determined by traditional methods and was compared with the instantaneous activation energy. It was found that while the activation energy changed with the progress of reaction over traditional methods, the instantaneous activation energy did not show any changes / or remained the same. The instantaneous activation energy was also found to be higher compared to the apparent activation energy determined with traditional methods.
Downloads
Published
How to Cite
Issue
Section
License
Authors retain copyright of the articles published in RILEM Technical Letters and grant the journal the right of first publication with open access. The work is simultaneously licensed under Creative Commons Attribution 4.0 International License (CC BY 4.0) that allows others to share and adapt the work under the following terms: 1) a proper attribution is given in a form of bibliographic record with the DOI link directing to RILEM Technical Letters; 2) a link to the license is provided; 3) the changes (if any) are indicated.









