Comparing the reactivity of different natural clays under thermal and alkali activation
DOI:
https://doi.org/10.21809/rilemtechlett.2019.85Abstract
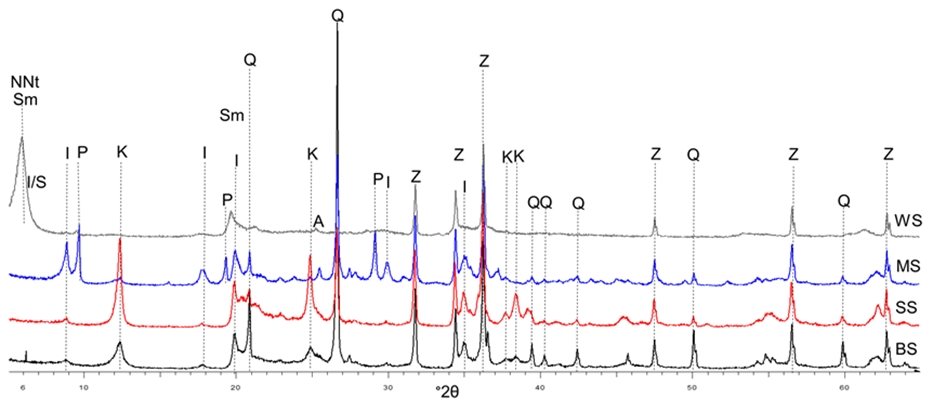
Metakaolin is one of the most popular solid aluminosilicate precursors for the synthesis of geopolymers. Despite its high reactivity and availability, there is a noticeable move towards the use of other natural clays as alternative precursors, due to their plentiful supply and widespread availability. Natural clays usually consist of a combination of 1:1 and 2:1 layer silicates reactivity of which vary. In this work, four different natural clays (SS, BS, MS and WS) composed of 1:1 and 2:1 clay minerals at different proportions were studied for the synthesis of geopolymers. To increase their reactivity prior to alkali activation, the clays were calcined in a laboratory oven at different temperatures (700, 750, 800, 850, 900 °C) and different holding times (10 seconds to 60 minutes). The reactivity of the calcined clays was assessed by the dissolution test and isothermal conduction calorimetry. NaOH and a mix of NaOH and Na2SiO3 were used as alkaline activators. The results show that the optimum calcination temperature of SS, BS and MS is 800 °C with a holding time of 10 minutes. WS is sufficiently calcined at a higher temperature of 900 °C for 20 minutes. Kaolinite-rich clays (SS) present more reactivity towards alkali activation than clays dominated by smectite or illite.
Downloads
Published
How to Cite
Issue
Section
License
Authors retain copyright of the articles published in RILEM Technical Letters and grant the journal the right of first publication with open access. The work is simultaneously licensed under Creative Commons Attribution 4.0 International License (CC BY 4.0) that allows others to share and adapt the work under the following terms: 1) a proper attribution is given in a form of bibliographic record with the DOI link directing to RILEM Technical Letters; 2) a link to the license is provided; 3) the changes (if any) are indicated.









